Nuclear Radius
Q7.
(a) Scattering experiments are used to investigate the nuclei of gold atoms.
In one experiment, alpha particles, all of the same energy (monoenergetic), are incident on a foil made from a single isotope of gold.
(i) State the main interaction when an alpha particle is scattered by a gold nucleus.
[1 mark]
(ii) The gold foil is replaced with another foil of the same size made from a mixture of isotopes of gold. Nothing else in the experiment is changed.
Explain whether or not the scattering distribution of the monoenergetic alpha particles remains the same.
[1 mark]
(b) Data from alpha-particle scattering experiments using elements other than gold allow scientists to relate the radius R, of a nucleus, to its nucleon number, A.
The graph below shows the relationship from the data obtained from a scattering experiment. The data obtained obeys the relationship R = r0 A1/3
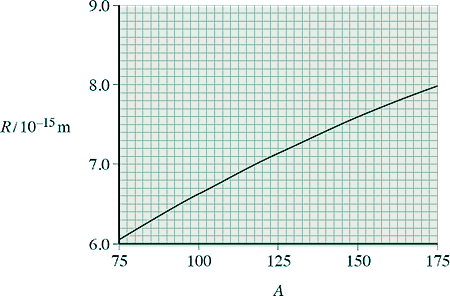
(i) Use information from the graph to show that r0 is about 1.4 × 10–15 m.
[1 mark]
(ii) Show that the radius of a  nucleus is about 5 × 10–15 m.
nucleus is about 5 × 10–15 m.
[2 marks]
(c) Calculate the density of a  nucleus. State an appropriate unit for your answer.
nucleus. State an appropriate unit for your answer.
[3 marks]
(Total 8 marks)



