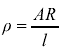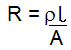Resistivity We discovered that -
At A level we put these into an equation:
Click here for some practice questions Here is an interactive Java application where
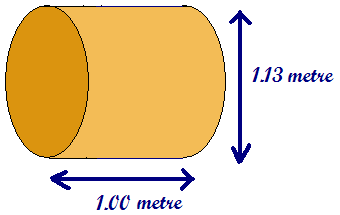
Unit of resistivity We can discover the unit for resistivity from this equation The unit of resistance multiplied by the unit for CSA divided by the unit for length That gives us Wm as the unit for resistivity. The electrical resistivity, or specific resistance, is the resistance between the opposite faces of a metre cube of a material. We are used to thinking of resistance in wires. So, it would be the resistance of a metre of wire with a cross sectional area of 1 m2 Imagine a wire like that!
Wow! What dimensions, hardly a wire at all - more like a metal cylinder! You would expect the resistivity of such a wire to be very small as the cross sectional area is so great.... and the values for resistivity of metals are very small. Nichrome
is quoted to have a resistivity of 103 X 10-8 Values are affected by impurities. Values given in different sources vary considerably. Resistivity is temperature dependent. The reciprocal of
the electrical resistivity is the electrical conductivity Click here to go to an excellent flash graphic of the periodic table and resistivity of elements! ----------------------------------------------- We can manipulate the equation to get R on the left
This equation is of the form Y=mx; it forms a straight line that goes through the origin. (See graphs section).
- If
your variable 'x' is the length then m (the gradient) becomes - If
your variable 'x' is 1/A then m (the gradient) becomes
G.W.C. Kaye and T.H. Laby in Tables of physical and chemical constants, Longman, London, UK, 15th edition, 1993. |
Follow me...
|