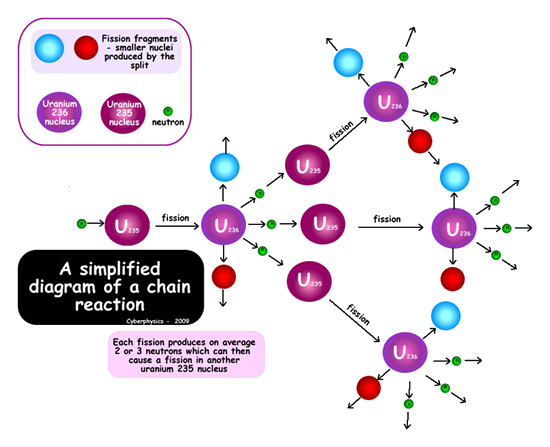
Chain reaction
Because a neutron is needed to instigate a fission (splitting) and neutrons are produced as the product of a fission, these products can go on to produce more fissions. The neutrons produced from these can then go on to produce more fissions etc.etc. Each stage is therefore linked to the previous one and therefore is termed a chain reaction. The only problem with the chain reaction is the fact that each stage of the reaction instigates more than twice as many fissions as the next stage (almost three times as many!).This results in the amount of energy produced at each stage more than doubling if the reaction is left unchecked. Each stage occurs in less than a millionth of a second therefore the heat energy produced can be phenomenal.The reaction is said to escalate. If we requires steady energy output of energy only one neutron from each fission should be allowed to go on to make another fission reaction. The reaction needs to be controlled - see control rods. The diagram below summarises what happens in a chain reaction. You are often asked to sketch a diagram to show chain reactions in exams - make sure you label them properly.
|
Follow me...
|