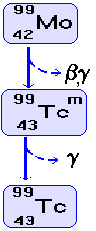Nuclear
Radiation from Human Activity
Nuclear
Power
Production of artificial radioisotopes
Alchemists dreamt of changing one element into another. With the advent of nuclear reactors that
is no longer just a dream! It is called artificial
transmutation of elements and it has allowed us to 'make' elements that
might have never before existed on our planet - the transuranic
elements!
Nuclear fission reactors
are not just a source of heat for power production. They are also an abundant
source of neutrons. As neutrons have
no charge, they have the ability to insert themselves in the nuclei
of a wide variety of elements, sometimes
turning the material into a radioactive
isotope of the original material that will then
decay into another element.
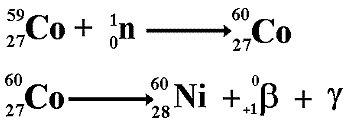
The main world isotope suppliers are
 Curium (France & USA),
Curium (France & USA),
 MDS Nordion (Canada),
MDS Nordion (Canada),
 IRE (Europe),
IRE (Europe),
 NTP (South Africa),
NTP (South Africa),
 Isotop-NIIAR (Russia), and
Isotop-NIIAR (Russia), and
 ANM (ANSTO Australia).
ANM (ANSTO Australia).
Most medical radioisotopes made in nuclear reactors are sourced from relatively few research reactors, including:
 HFR at Petten in Netherlands (supplied via IRE and Curium).
HFR at Petten in Netherlands (supplied via IRE and Curium).
 BR-2 at Mol in Belgium (supplied via IRE and Curium).
BR-2 at Mol in Belgium (supplied via IRE and Curium).
 Maria in Poland (supplied via Curium).
Maria in Poland (supplied via Curium).
 Orphee at Saclay in France (supplied via IRE).
Orphee at Saclay in France (supplied via IRE).
 FRJ-2/FRM-2 at Jülich in Germany (supplied via IRE).
FRJ-2/FRM-2 at Jülich in Germany (supplied via IRE).
 LVR-15 at Rez in Czech Republic (supplied via IRE).
LVR-15 at Rez in Czech Republic (supplied via IRE).
 HFETR at Chengdu in China.
HFETR at Chengdu in China.
 Safari in South Africa (supplied from NTP).
Safari in South Africa (supplied from NTP).
 OPAL in Australia (supplied from ANM).
OPAL in Australia (supplied from ANM).
 ETRR-2 in Egypt (forthcoming: supplied to domestic market).
ETRR-2 in Egypt (forthcoming: supplied to domestic market).
 Dimitrovgrad in Russia (Isotop-NIIAR).
Dimitrovgrad in Russia (Isotop-NIIAR).
Depleted
Uranium (DU)
Depleted uranium is
a by-product of the nuclear fuel industry.
It gets its name because it's
been stripped, or depleted, of most of its content of Uranium 235 - which
is used as fuel in nuclear reactors. It is primarily uranium 238 - an alpha emitter.
Depleted uranium is
used in the production of anti-tank weapons and bullets because it's an
extremely dense metal; a shell tipped with DU can rip through a heavily
armoured tank! There is considerable controversy over whether it is safe
or ethical to use such shells.
Many media sites
have considerable information on this but it arouses strong feelings and
therefore many of the accounts will be biased one way or the other. Always
look at who wrote an article and think why they did it. Also always try
to find a range of views and look for use of good sound scientific principles
in any argument.
Disposal
of nuclear waste
High
Level Waste
This waste requires very
heavy shielding as it is very radioactive.
The intense radioactive decay
generates a large amount of heat. this needs to be carefully considered
when thinking about storage and final disposal.
High Level Waste includes
spent fuel and highly radioactive liquids generated during reprocessing
operations. The latter is all stored at Sellafield in high-integrity stainless
steel tanks fitted with cooling coils to remove the heat generated. Management
and disposal of this waste is difficult due to the high levels of radioactivity,
the very long half-lives of some of the radionuclides present and the
heat continually generated as a result of decay processes.
Current practice
is to store these wastes, encapsulated in glass, in air cooled steel containers
for 50 years to allow the heat generated to reduce to manageable levels,
and then finally dispose of them in a deep mine.
Intermediate
Level Waste
This waste also needs
to be heavily shielded, as it can be extremely radioactive, but does not
generate as much heat as High Level Waste. Some of the radioactive particles
present in this waste may have very long half-lives and so require isolation
for many thousands of years.
Intermediate Level
Waste includes fuel element claddings removed prior to reprocessing, various
sludges and ion exchange resins from fuel storage pond water treatment;
concentrates of liquid waste streams; heavily contaminated scrap equipment;
plutonium contaminated materials and graphite sleeves and steel components
from AGR fuel assemblies. Large volumes of Intermediate Level Waste are
expected from decommissioning operations are expected in the coming years.
Because of the wide
range of Intermediate Level Waste sources many different forms of conditioning
and packaging are required.
Low
Level Waste
This waste tends to be
low in radioactivity and high in bulk. It ranges from general rubbish (gloves,
clothing, packaging, paper towels, over shoes, laboratory glassware, etc.)
to some very low-level plutonium contaminated materials.
A lot of material
classified as Low Level Waste, may in fact not be radioactive at all,
but it is potentially radioactive through being in an active/contaminated
area.
The low levels of
radioactivity and the short-lives of the contaminants mean this waste
is relatively harmless if handled properly. However, any site used for
Low Level Waste disposal will need to be subject to land use restrictions
for around 300 years after the site is closed. There is also always a
risk of environmental problems if water leaching through the waste site
finds its way into surface and ground waters.
Medical
uses of nuclear radiation
Radioactive
tracers
A
radioactive tracer is very
useful to doctors. A small amount of radioisotope
is made to replace a non-radioactive isotope of an element
in a compound that normally performs
a task in the body. The path of that radioisotope or one of its daughter
nuclei (product of the decay) is then monitored by detecting the emitted
nuclear radiation.
The type of
radiation emitted by a medical tracer is very important.
Gamma emitters are ideal because of their
low ionizing power. But gamma emission
often follows closely on alpha or beta
emission making the isotope unsuitable. Ideally we need to find a pure
gamma emitter (one with a metastable nucleus).
The nucleus should not be likely to emit α or β after emission of the γ-ray either
so decay to the next atom in the radioactive
decay series should involve a long half-life.
The energy of the γ-ray emitted should be suitable
for viewing with a gamma-camera
(energy range 100 - 400 keV gives optimum
detection, is easy to collimate
and has low attenuation in the body).
For example:
Technetium
99m
Technetium
99m is a very useful radioisotope. It is the most commonly used one
in hospitals because it is ideal to use with a gamma camera and only
emits gamma rays.
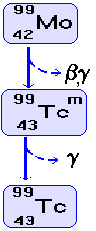
Technetium 99m is a very useful radionuclide
in gamma imaging because:
 It only emits gamma rays and
these are of an energy that is easily detected by a gamma camera
(140keV).
It only emits gamma rays and
these are of an energy that is easily detected by a gamma camera
(140keV).
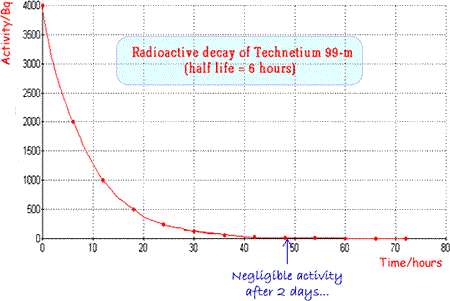
 It has an ideal half life
(six hours) which is long enough for diagnostic procedures to take
place but short enough for the patient not to be inconvenienced
unduly by remaining 'radioactive'
for too long a period after the investigation. (See graph above)
It has an ideal half life
(six hours) which is long enough for diagnostic procedures to take
place but short enough for the patient not to be inconvenienced
unduly by remaining 'radioactive'
for too long a period after the investigation. (See graph above)
 It is suitable not only for use alone but also for attachment to
a wide range of compounds for tracer experiments.
It is suitable not only for use alone but also for attachment to
a wide range of compounds for tracer experiments.
 It
can easily be produced 'in situ' using a 'cow' as it is the
daughter nucleus of the decay
of Molybdenum 99 and is easily separated from the parent (more hazardous
beta-emitter) by a saline flush. A fresh supply is brought to the
hospital fortnightly and then the 'cow' is 'milked' as and when
required.
It
can easily be produced 'in situ' using a 'cow' as it is the
daughter nucleus of the decay
of Molybdenum 99 and is easily separated from the parent (more hazardous
beta-emitter) by a saline flush. A fresh supply is brought to the
hospital fortnightly and then the 'cow' is 'milked' as and when
required.
Gamma Camera
The gamma camera allows an image to be formed on a screen that shows how
intensely gamma rays are being emitted from a particular part of the
body.
A Technetium-99m antimony sulphide colloid can be used as a radioactive
marker to examine the function of the lymph nodes.
It
is carefully injected into the drainage areas to be visualised.
|
|
A 'butterfly'
(so-called because of its appearance) can be used to ensure that
an injection of radioactive material is inserted into the correct
site.
It allows the doctor or nurse to check that the needle is
inserted properly before the radioactive fluid is pumped into
the patient. The fluid needs to be injected into the blood stream. If it goes into tissue it will be concentrated in a small area instead of being distributed throughout the blood (a large volume!) - it could therefore cause tissue damage.
|
The
radionuclide in the syringe is shielded to protect the person giving
the injection.
It is important to minimise the dose of personnel working with radioactive materials as each time they are exposed to radiation they increase the odds on their contracting cancer at a later date. |
 |
(Above
two images from CALRAD an interactive educational package designed by
an educational consortium of several Universities - I came across it
via the Open University module S803)
The
gamma ray is electromagnetic radiation of very high penetration
power. Therefore more rays exit the body and are available for detection
than interact with the patient's tissue. The patient is now emitting
gamma rays!
The
doctor then gets the patient to wait for a while. This will give
the tracer-chemical a chance to accumulate in the parts of the body
the non-radioactive chemical would have done normally.
Cancerous
cells divide more frequently than non-cancerous ones and therefore a
'hot-spot' of high activity results
from any cancerous growth. A gamma camera is scheduled to scan
the area of interest approximately four hours after the patient has
been injected with the radioactive tracer. This gives it time to circulate
and accumulate in 'hot-spots' of rapid cell division. A second scan
is then taken within the 24-hour period and compared to the first to
diagnose suspicion of overactive lymph node activity.
PET
(Positron Emission Tomography)
PET
is an imaging technique with many uses. It can be used for:
 bone imaging,
bone imaging,
 monitoring
tumour metabolism,
monitoring
tumour metabolism,
 monitoring
the function of the heart : cardiac perfusion and myocardial blood-flow
monitoring,
monitoring
the function of the heart : cardiac perfusion and myocardial blood-flow
monitoring,
 studying
fatty acid metabolism
studying
fatty acid metabolism
It is not widely available
because the equipment is very expensive. It is found in research establishments and requires highly qualified staff to carry out scans.
When a positron is emitted it is soon annihilated and a pair of gamma
rays is produced.
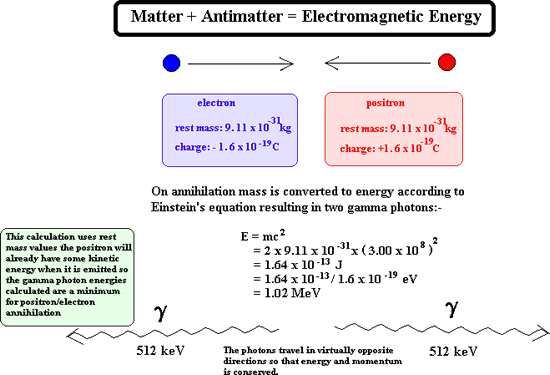
These gamma rays fly
off in opposite directions and can be viewed using a PET Scanner. This
is a special type of gamma camera that is designed to count only gamma
rays that are produced in pairs. It is linked to a computer that can deduce
where the annihilation of the positron took place.
Useful
URLs
There are many PET
sites on the WWW but care has to be taken when searching because of confusion
with 'pet animals'! (inclusion of the word 'gamma' usually overcomes this)
http://www.imaginis.com/nuclear-medicine/
http://www.nucmednet.com/frameset.htm
(this site has a
'real-time' image of a beating heart being monitored by gamma radiation)
The Gamma
Camera - Click here
Radiation
Therapy
 Tumours
can be treated using gamma or X-rays.
Tumours
can be treated using gamma or X-rays.
Cells that divide rapidly are more prone to damage by high-energy electromagnetic
radiation. This means that tumour cells are more radiosensitive
than their normal counterparts.
By carefully aiming the rays at the tumour
(gamma-ray beams directed from a multitude of angles that result in the
maximum gamma ray intensity within the tumour) the harmful effect of the ionising radiation is kept to a minimum
in the surrounding tissue.
This kind of treatment is most hazardous when
a brain tumour is being irradiated and the surrounding tissue is vital
for normal brain function.
Several treatments are usually given over a
time period of several weeks to minimise the unpleasant side effects (most
commonly nausea, sickness, and tiredness).
New methods of delivering radiation treatment have been developed
-
 Interstitial
radiation involves implanting radioactive
chemicals (termed seeds) directly into a tumour.
Interstitial
radiation involves implanting radioactive
chemicals (termed seeds) directly into a tumour.
 Stereotactic
radiosurgery delivers a high, single dose
of radiation to a small, well-defined area.
Stereotactic
radiosurgery delivers a high, single dose
of radiation to a small, well-defined area.
Useful URLs
http://www.times-archive.co.uk/JohnDiamond/
(An excellent resource for bringing home the 'human side' of treatments
- select the article published 22nd July 2000 - he describes
what its like on the receiving end of treatment requiring radioactive
wires inserted into the neck)
http://www.salu.net/sites/r/rtsideffects/
(general information on possible side effects of treatment)
http://www.radiotherapy.com/education/index.htm
http://www.oncolink.com/specialty/rad_onc/general/xrt_intro.html
http://www.oncolink.com/pdq_html/6/engl/600071.html
http://www.biomed.org/pet.html#what
http://www.valley-radiotherapy.com/treatment/specific/index.asp



bone imaging,
monitoring tumour metabolism,
monitoring the function of the heart : cardiac perfusion and myocardial blood-flow monitoring,
studying fatty acid metabolism

 Tumours
can be treated using gamma or X-rays.
Tumours
can be treated using gamma or X-rays. ![]() Interstitial
radiation involves implanting radioactive
chemicals (termed seeds) directly into a tumour.
Interstitial
radiation involves implanting radioactive
chemicals (termed seeds) directly into a tumour.![]() Stereotactic
radiosurgery delivers a high, single dose
of radiation to a small, well-defined area.
Stereotactic
radiosurgery delivers a high, single dose
of radiation to a small, well-defined area.